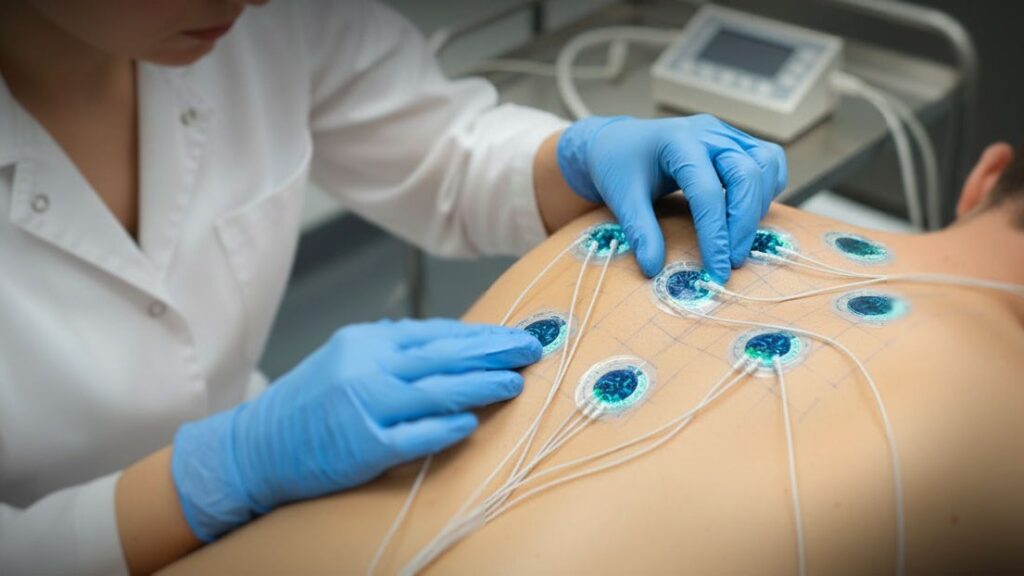
Medical sensors sit at the intersection of engineering precision and patient safety. Whether they are monitoring heart rhythms, measuring muscle stimulation, or capturing bio signals in real time, there is very little tolerance for variability.
Yield is not just a production metric in sensor manufacturing. It is a reflection of process control, material discipline, and manufacturing maturity.
In my experience working alongside engineering teams and sourcing leaders, the conversations are rarely about just price. They are about stability. Repeatability. Confidence. The question is not whether a sensor can be built. The question is whether it can be built consistently, at scale, without surprises.
At Printec, yield and risk mitigation are treated as connected priorities. One cannot improve without addressing the other.
Medical Sensor Manufacturing Yield Depends on Process Control
Unlike many mechanical components, sensors are sensitive to subtle variations. Adhesive thickness, conductive ink consistency, lamination pressure, curing cycles, and environmental conditions all influence final performance.
Small process deviations can result in signal drift, inconsistent readings, or early field failures. These are not issues that show up immediately. They reveal themselves later, often at the worst possible time.
For engineers, this means every variable must be understood and controlled. For procurement leaders, it means supplier process discipline directly impacts long term program risk.
Common Threats to Medical Sensor Manufacturing Yield
Most yield loss does not come from dramatic failures. It comes from incremental inconsistency.
Material variability between lots Inadequate environmental control during printing or assembly Insufficient in process inspection Manual steps without validation safeguards
Each of these may seem manageable in isolation. Together, they compound.
When yield drops even a few percentage points, total program cost rises quickly. Scrap increases. Lead times extend. Rework consumes engineering resources. Forecasts become unstable.
Improving yield is not just about saving material. It is about protecting the entire program.
Building Process Visibility Instead of Reacting to Defects
One of the most effective ways to reduce risk in medical sensor manufacturing is to shift from reactive quality to proactive process monitoring.
This includes structured incoming material verification, documented process parameters, and in process inspection checkpoints that validate critical characteristics before final assembly. Process controls should align with ISO 13485 quality management system requirements.
When issues are detected early, correction is controlled and contained. When issues are detected late, they become expensive.
For engineers, this creates a more predictable validation pathway. For buyers, it reduces the likelihood of sudden disruptions or cost escalations.
The Real Cost of Poor Medical Sensor Manufacturing Yield
It is easy to view yield purely as a factory metric. In reality, it influences far more.
Lower yield affects delivery commitments. It impacts supplier credibility. It drives unexpected cost increases. It introduces regulatory exposure in tightly controlled programs.
Strong yield performance signals something deeper. It signals process maturity.
Organizations that invest in process control, documentation discipline, and continuous improvement are not simply manufacturing parts. They are protecting their customers from unnecessary risk.
Closing Perspective
Medical sensor manufacturing demands more than capability. It demands control.
Reducing risk and improving yield are not separate initiatives. They are outcomes of disciplined systems, engineering alignment, and consistent execution.
When processes are stable, sensors perform reliably. When sensors perform reliably, trust follows.
And in medical manufacturing, trust is everything.